Guidelines for the Management of Patients With EoE
The American Gastroenterological Association (AGA) and the Joint Task Force on Allergy-Immunology Practice Parameters (JTF) have published guidelines for the management of patients with eosinophilic esophagitis (EoE).1
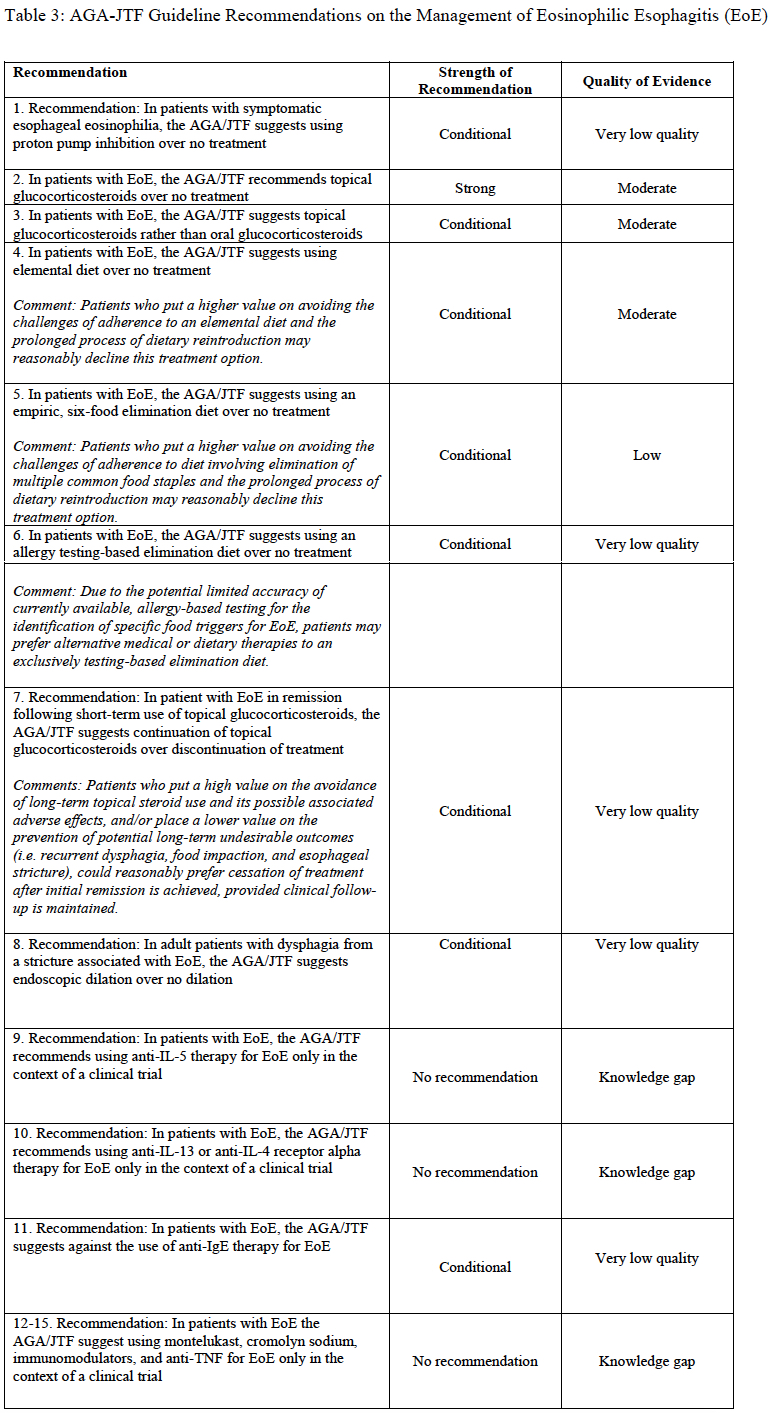
Among the fifteen total recommendations, there was one strong recommendation, there were eight conditional recommendations, and three “no recommendations.” The strong recommendation was for topical glucocorticosteroid use over no treatment for patients with EoE.
To find out more about the recommendations, Consultant360 reached out to the Chair of the Writing Committee, Ikuo Hirano, MD, who is a professor of medicine in the Division of Gastroenterology and Hepatology at Northwestern University Feinberg School of Medicine in Chicago, Illinois.
Consultant360: What was the impetus for these guidelines?
Ikuo Hirano, MD: EoE was first characterized as a distinct clinical entity in 1993 and thought of as an esoteric condition at the time. EoE is now widely recognized as a common cause of esophageal morbidity and a leading cause of dysphagia in adults. These guidelines represent the first recommendations endorsed by the AGA Institute and JTF that address this important clinical condition.
C360: The guideline includes fifteen recommendations, with only one strong recommendation for the treatment of patients with EoE. How were the eight conditional recommendations and three “no recommendations” determined?
Dr Hirano: The AGA-JTF guidelines followed the GRADE approach that considers not only the strength of the evidence but also the balance between desirable and undesirable consequences of an intervention, as well as the values and preferences of patients. Conditional recommendations were made due to limitations in the evidence base and qualifications based on patient preferences. We expect that the recommendations will evolve as research continues in this relatively new disease.
C360: The only strong recommendation made in the guideline is for use of swallowed topical glucocorticosteroids over no treatment. Which treatment options are currently available? Which novel therapies, such as biologics, are currently being investigated?
Dr Hirano: There are currently no medications approved by the US Food and Drug Administration (FDA) for the treatment of EoE. The European Medicines Agency recently approved an orally administered tablet formulation of budesonide for EoE that is not available in the United States.
While awaiting the approval of therapies by the FDA, treatment options at this time include the off-label use of swallowed topical glucocorticosteroids (developed for asthma but repurposed for EoE), proton-pump inhibitors, and elimination diets. Esophageal dilation is an effective option for EoE patients with dysphagia secondary to esophageal strictures. Novel therapies in ongoing clinical trials include liquid and tablet formulations of glucocorticosteroids designed to optimize esophageal delivery, as well as biological therapies that target allergic/immune pathogenesis including the IL-4, IL-13, and Siglec-8 receptors.
C360: Do the recommendations differ for children vs adults?
Dr Hirano: The patient populations in the EoE recommendations are inclusive of both children and adults with EoE. It is important, however, to recognize that the evidence base informing the recommendations are variably based on primarily pediatric or adult studies. Such qualifications are noted in the comments that follow each recommendation and in the technical review. In addition, the use of esophageal dilation is most commonly applied to adults with EoE, since esophageal strictures are less prevalent among children.
C360: There seems to be a lack of quality evidence regarding the efficacy of treatment options for patients with EoE. Are there any ongoing studies to fill the knowledge gaps and better inform treatment decisions?
Dr Hirano: As a relatively new disease, there are many knowledge and evidence gaps in the management of EoE. Several of these gaps are highlighted in a table in the guidelines. The increasing number and quality of clinical trials in EoE over the past decade will undoubtedly address these gaps and improve patient-care decisions. (Click table to enlarge.)
Reference:
- Hirano I, Chan ES, Rank M, et al; the AGA Institute Clinical Guidelines Committee and the Joint Task Force on Allergy-Immunology Practice Parameters. AGA Institute and the Joint Task Force on Allergy-Immunology practice parameters clinical guidelines for the management of eosinophilic esophagitis. Gastroenterology. 2020;158(6):1776-1786. https://doi.org/10.1053/j.gastro.2020.02.038


